
100 Years, 100 Breakthrough T1D Scientists: The BB Rat—An Animal Model of Type 1 Diabetes
In 1974, a colony of non-diabetic BB rats develops diabetes. Examination of their pancreases revealed that they had no beta cells.

Employee Spotlight: Meet Tricia Martin
This Black History Month, the Black at Breakthrough T1D Employee Resource Group, in collaboration with the Diversity and Inclusion Task Force and the Greater New York City Chapter, would like to acknowledge our colleague Tricia Martin whose more than 20 years of service to Breakthrough T1D exemplifies Black Excellence and a commitment to diversity and […]

The First Gene-Edited Cell Replacement Therapy Takes a Step Forward
The first person has received ViaCyte and CRISPR's cell replacement therapy.
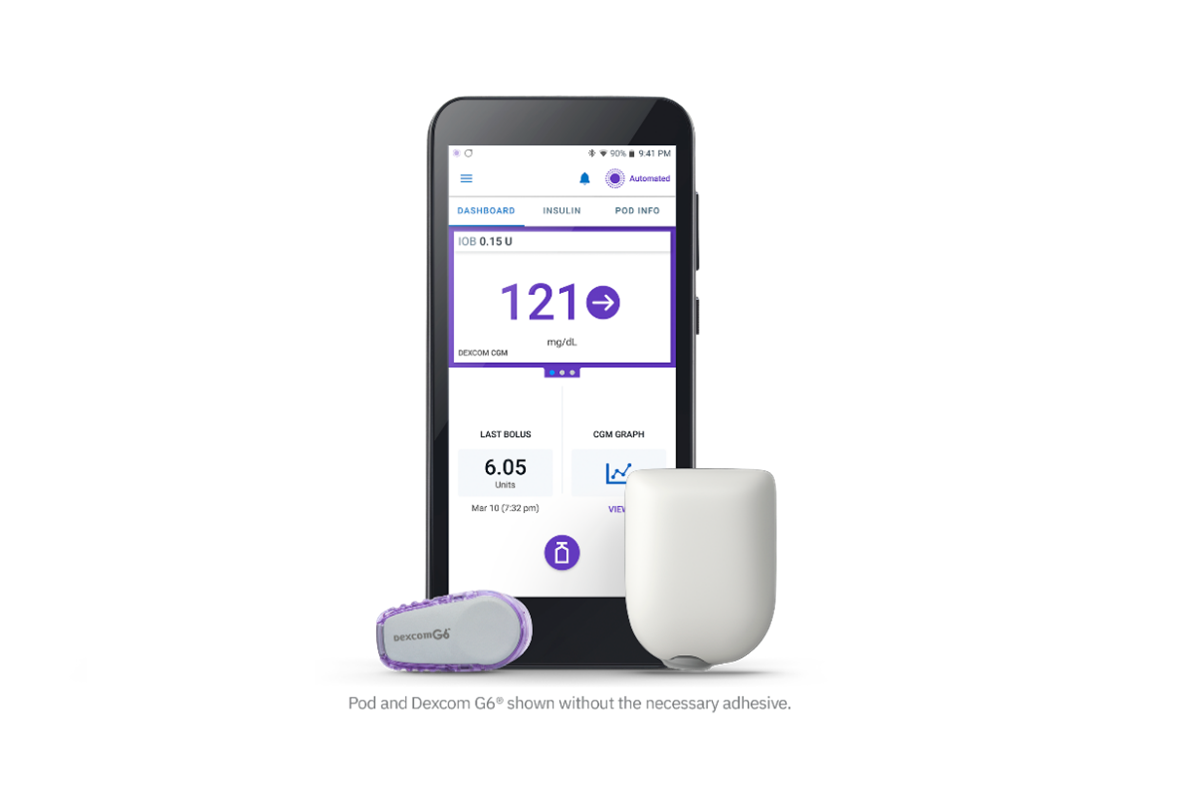
FDA Authorizes a Fourth Artificial Pancreas System
The FDA authorized the Insulet Omnipod 5, the world’s first tubeless, wearable system for people 6 and older, using an algorithm that began by Breakthrough T1D funding.

Interview with our New Community Screening and Clinical Trial Education Director
Breakthrough T1D has a new community screening and clinical trials education director. She's been a volunteer and now staff since her daughter was diagnosed in 2002.

New Study Shows Kids 1+ Benefit from Artificial Pancreas Technology
A new paper published in the New England Journal of Medicine shows hybrid closed loop systems help children as young as 1 year old achieve better glycemic control.

100 Years, 100 Breakthrough T1D Scientists: Gian Franco Bottazzo, M.D.
In honor of the 100th anniversary of insulin, we are launching “100 Years, 100 Breakthrough T1D Scientists.” The first up: Gian Franco Bottazzo, M.D.

Rufus, the Bear with Diabetes®, “2.0”
He’s cute. He’s fuzzy and cuddly. And thanks to the Breakthrough T1D-Beyond Type 1 Alliance, Rufus, the Bear with Diabetes® has gone through a major upgrade!
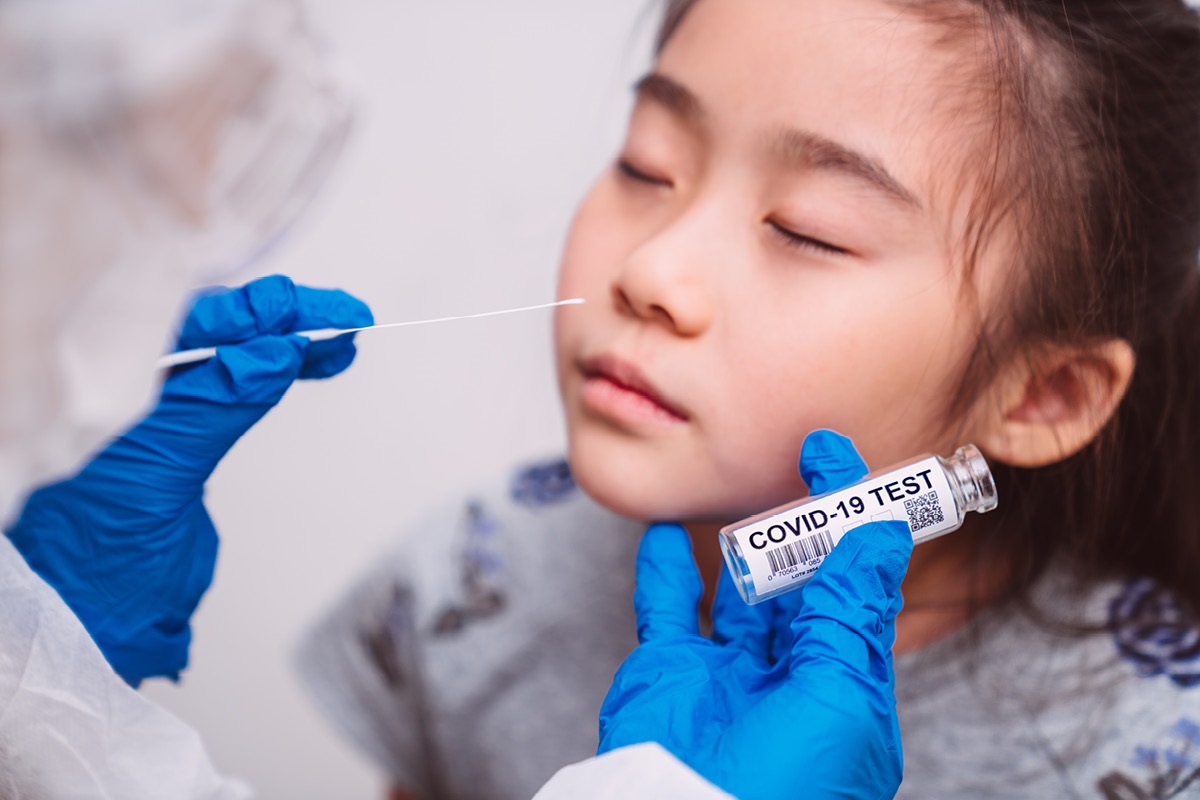
CDC Study: Children Who Have Recovered from COVID-19 May Be at Increased Risk of Diabetes
CDC researchers report an apparent association between pediatric COVID-19 and diabetes diagnoses.

Insulin: A Story of Innovation
On January 11, 1922—100 years ago—14-year-old Leonard Thompson, who was diagnosed with diabetes 3 years earlier, became the first person to receive insulin.